Medical devices and Healthcare: accelerate innovation and meet ever-changing regulations
Software has become a real business asset for manufacturers of medical devices. They can benefit from endless opportunities to innovate. However, the MedTech is a highly regulated industry that requires quality to be the cornerstone of product development.
With Tuleap, you can now build safe and high quality products and achieve proof of compliance with minimal effort.

Main Challenges in Medical Device Development

Accelerate delivery of innovative medical technology
Make software innovation at the heart of your medical device and keep up with competitors. Let’s unlock agile’s potential to increase your teams’ productivity and gain benefits for the development of medical devices: adaptability and flexibility of your teams over time, fast ROI with continuous improvement, risk reduction thanks to consistent testing and higher software quality.
Achieve proof of compliance, faster
As the Medical industry faces increasingly strict regulation, it is crucial that the medical device design, construction, sales and maintenance respect several standards to ensure your medical device reliability and sustainability on the market. Amongst the standards, these two down below drive the daily work of the development teams:
- The ISO 13485 standard ensures the quality and efficiency of processes while continuously improving the products reliability on the market thanks to a rigorous quality management system (QMS).
- The IEC 62304 standard defines the requirements for the overall software development cycle of medical devices.
How Tuleap helps companies to develop quality Medical Devices
Implement Agile and DevOps best practices
Provide full traceability from initial requirements to tests and deliveries
Bring business analysts, developers, testers, and quality managers together on the same workspace
Automate workflows and Proof of Compliance
Prepare and pass audits easier, faster, for a cost-effective compliance
Trusted by innovative organisations in the Medical industry


Tuleap brings greater security, efficiency, and trust to our audit-related processes for ISO 13485 compliance.
Nicolas GAIFFE, Software Development Manager, Sleepinnov Technology
Tuleap Capabilities for Medical Device Software Development
1. Agile and Compliance made possible together
Despite some concerns, agile approaches are totally suitable for developing medical device software. Agility integrates regulatory compliance and medical device development, enabling organizations to develop compliant medical devices. With Tuleap agile project management tools, discover the power of Agile and DevOps principles to successfully develop high-quality medical devices (MD):
- Better collaboration among teams,
- Short iterations and adaptive scheduling,
- Continuous deployment and delivery,
- Flexible response to changing circumstances,
- Early testing.


2. Medical Device Requirement and Risk Management
Rigorous risk management is a crucial pillar to meet the medical industry standards and prove your compliance. This is why Tuleap integrates an Issue Tracking module to design and set up a flexible, sophisticated and customized system that best adapts to your business challenges:
- Gather, organize, plan and track any functional and/or non-functional specifications throughout the medical device lifecycle
- Index risks and define mitigating measures to get an “acceptable” level of risk
- Create tailor-made requirements approval and validation processes with advanced workflow automations
- Generate test cases based on requirements
3. Embedded Software Development and Baselines
Enhance the robustness of your medical products by delivering high quality software.
- Leverage a Continuous Development chain with Change Management, Version Control & Baselines in one place.
- Provide end-to-end traceability through code versioning, code review and development activities in customized collaborative workspaces.
- Define and reuse mature and proven development processes to ensure compliance with access control.
- Link easily regulatory requirements to development artifacts, and trace specifications down to source code.
- Save, browse, and compare requirement baselines across versions.


4. Automated Traceability for faster medical audits
To limit your exposure to regulatory risks, such as non-conformity ones, you need a traceable record or log as to be able to prove that you are 100% compliant. Tuleap ensures gap-free traceability throughout the entire product development lifecycle providing a large set of integrated tools.
- Collect, organize, and manage all required evidence in one single place as a source of truth,
- Achieve end-to-end product traceability, tracking changes on any medical project artifacts and components across the value chain, including the design, engineering, delivery, and service phases,
- Create corporate templates to standardize validated processes, reduce the number of errors by reusing project configurations and verified processes,
- Cut the costs in terms of time and money for audit preparation with quick, easy access to complete product history information, conveniently located in one place.
5. Medical Quality Assurance and Testing
- Accelerate product validation with Tuleap Test Management™ module, which is directly integrated with other Tuleap tools,
- Use fast, efficient, and accurate automated and manual testing to validate your software products,
- Track and manage all products’ quality assurance activities from requirements to releases, all this in one single Tuleap interface,
- Launch, monitor and analyze test campaigns in real time.

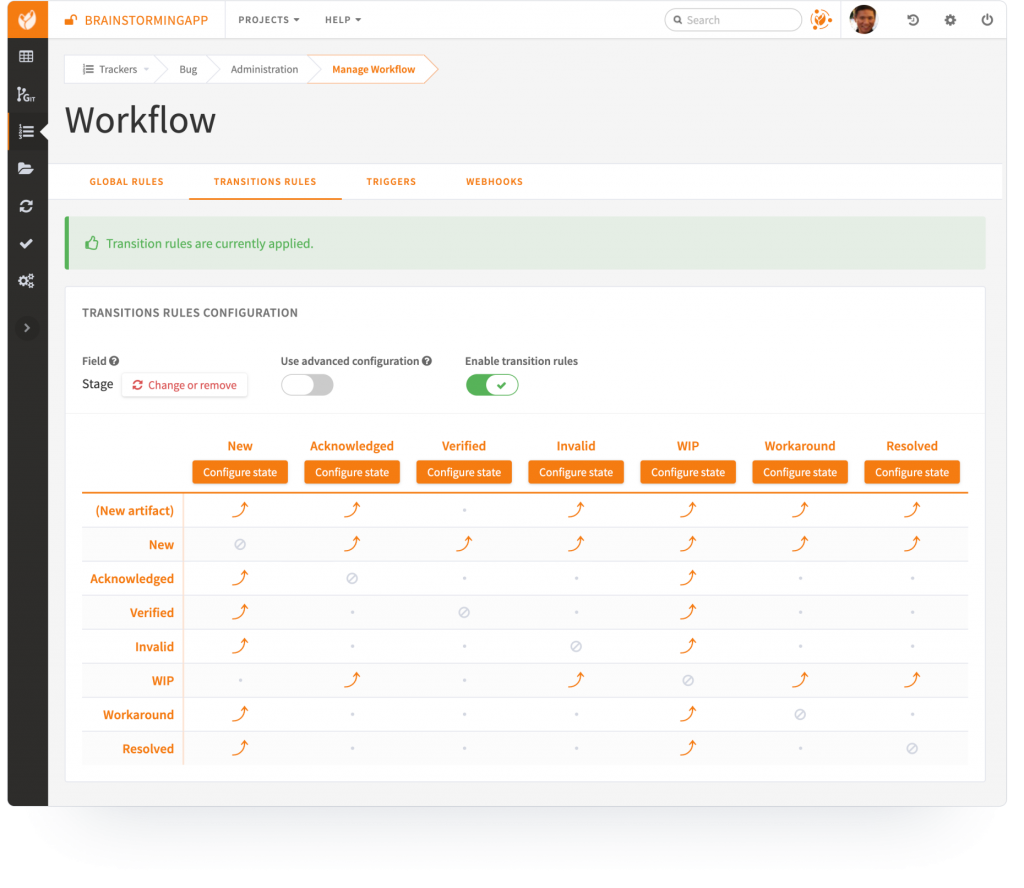
6. Workflow automation and Quality Management System
Tuleap Issue Tracking™ module is therefore highly adaptable to your development and quality management policies:
- Configure and enforce workflows in the product development process thanks to advanced setups and triggers,
- Define conditional actions to be triggered by specific events,
- Manage access permissions to certain medical device projects and confidential information thanks to a sophisticated authentication system
7. Document Generation and Management
- Generate requirements specification documents in a few seconds and export any Test Plan information in a document form (requirements, tests, campaigns, execution status and so on),
- Get a traceability matrix to easily prove your products’ quality and standards compliance (i.e. ISO 13485, IEC 62304…),
- Share quality-related documentation internally or externally via Tuleap Document Manager™ : create, update, review, track changes and approve documents,
- Prevent the use of obsolete documents and ensure error-free archiving,
- Set document access permissions for a secure workspace and implement user- and role-based features for permission control.

Ready-to-use template for Software Medical Device projects
The Tuleap Template for MedTech Developments contribute to better manage the entire lifecycle of software products as well as achieve compliance easier and faster by proving the quality of your delivered products.
The template help you meet your business challenges such as ensuring full traceability, implementing a Quality Management System to both improve your medical device management across teams and comply with the ever-evolving industry standards.

What MedTech companies say about Tuleap

Open Source Business Partners for the long haul – the kind of people we want to work with.
Portfolio Management -Healthcare – Nov, 2021
Great user-friendly poject management tool.
Portofolio Management – Healthcare Industry – Nov 2021
Very good product, intuitive, easy to walk through and versatile.
Portofolio Management – Healthcare Industry – Nov 2021
Very powerful tool that brings the project management at a higher level. A must have.
R&D Development – Healthcare- Nov, 2021
Replay the webinar
ISO 13485 – IEC 62304: Keep processes under control to make audits easier
It is time for action now! Let’s provide teams with the right tools to define and optimize the management of medical device (MD) projects and show proof of the quality of your software for audit compliance purposes.

